Understanding the ExoDisc Advantage: What the Research Reveals
A groundbreaking study published in Lab on a Chip (2024) validates ExoDisc as a transformative platform for both exosome isolation and therapeutic cargo loading. The research demonstrates that ExoDisc's proprietary tonicity control (TC) method enables rapid, efficient loading of diverse therapeutic molecules—including chemotherapeutic agents, nucleic acids, and proteins—into extracellular vesicles without compromising their structural integrity or biological function.
The study's key findings establish ExoDisc's clinical utility: the platform achieves 4.3-fold higher loading efficiency compared to sonication and 7.2-fold improvement over extrusion methods. These aren't marginal gains—they represent paradigm-shifting performance that directly impacts therapeutic development timelines and research outcomes. When laboratories process patient samples or conduct therapeutic development work, these efficiency multipliers translate to substantial cost savings and accelerated time-to-insight.
The Science Behind Superior Performance
ExoDisc leverages centrifugal tangential flow filtration through anodic aluminum oxide membranes with precisely engineered 20 nm pores. This design enables label-free, highly efficient isolation of extracellular vesicles from biological samples including cell culture supernatants, plasma, and other body fluids. The platform's innovation extends beyond isolation—its integrated tonicity control methodology represents a breakthrough in cargo loading technology.
The TC approach operates through controlled membrane permeabilization using hypotonic solutions (optimized at 296 μOsm L⁻¹), temporarily increasing membrane permeability to allow therapeutic cargo influx, followed by isotonic washing that restores membrane integrity and removes unencapsulated materials. This gentle, chemical-free process preserves essential exosome characteristics including surface markers (CD9, CD63, CD81, TSG101), morphology, and functional properties—critical factors for downstream applications.
Dynamic light scattering analysis confirms that while exosomes temporarily swell during hypotonic exposure (26.8% size increase), they return to original dimensions after isotonic restoration with no permanent structural changes. Transmission electron microscopy imaging verifies intact double-layer membrane architecture post-processing. Western blot analysis, ELISA testing, and qRT-PCR evaluation of mRNA markers demonstrate complete preservation of protein expression and internal components—validation that ExoDisc processing maintains exosome biological authenticity.
Clinical Workflow Integration: Designed for Real-World Laboratory Environments
For clinical laboratories evaluating exosome isolation technologies, workflow compatibility often determines adoption success more than theoretical performance metrics. ExoDisc addresses practical implementation barriers through intelligent design choices that respect laboratory realities.
Benchtop Operation with Standard Equipment: Unlike ultracentrifugation requiring specialized, expensive ultracentrifuges occupying significant laboratory footprint, ExoDisc operates on standard benchtop centrifuges already present in most clinical laboratories. The platform spins at 3,000 rpm—conventional speeds accessible to general-purpose centrifuges, eliminating capital equipment barriers and extensive operator training requirements.
Rapid Processing for Clinical Timelines: The complete isolation and cargo loading workflow executes in approximately 15 minutes, consisting of three 5-minute centrifugation cycles. This rapid turnaround enables same-day processing of clinical samples, supporting timely diagnostic decisions and research workflows. Contrast this with ultracentrifugation protocols requiring 6-24 hours, or multi-step precipitation methods involving overnight incubations—timelines incompatible with urgent clinical needs.
Minimal Sample Transfer and Particle Loss: ExoDisc's integrated design performs isolation, cargo loading, and washing on a single disc platform without sample transfer between vessels. This continuity minimizes handling steps that introduce contamination risks and particle losses. The published research demonstrates zero significant particle loss with ExoDisc TC processing, compared to 34.9% loss with sonication and 57.0% loss with extrusion methods. For precious clinical samples where material quantity limits analytical depth, preservation of every available vesicle matters substantially.
Scalability and Reproducibility: The platform's closed-system design and standardized protocols ensure reproducible results across operators, sample types, and laboratory settings. This consistency supports clinical validation studies, multicenter trials, and quality control requirements essential for diagnostic applications. The ability to process samples ranging from small research quantities to larger clinical volumes provides flexibility for varied laboratory needs.
Competitive Advantages: How ExoDisc Outperforms Traditional Technologies
Clinical laboratories currently employ several exosome isolation methodologies, each carrying distinct limitations that ExoDisc systematically addresses. Understanding these comparative advantages helps laboratories make informed technology selection decisions.
Performance Comparison Table
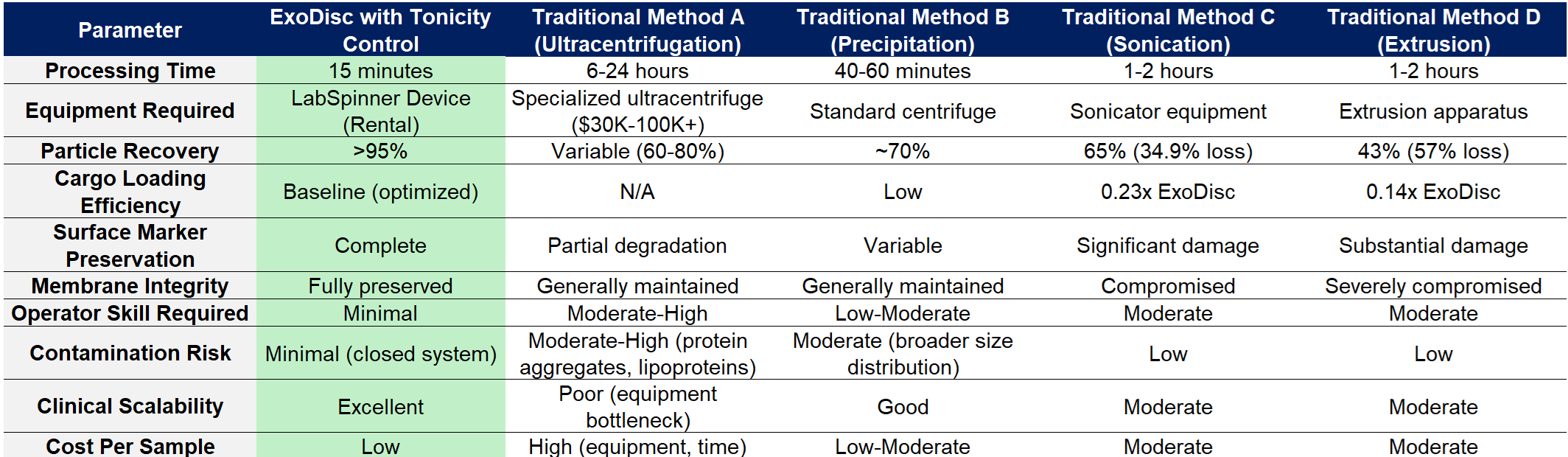
Ultracentrifugation: While historically considered the "gold standard," ultracentrifugation's practical limitations increasingly outweigh its theoretical benefits. The method requires 6-24 hour processing times incompatible with clinical decision-making timeframes. Specialized ultracentrifuge equipment demands significant capital investment ($30,000-$100,000+) and dedicated laboratory space. Protein aggregate and lipoprotein contamination compromises sample purity, potentially confounding downstream analyses. ExoDisc delivers superior isolation speed without sacrificing quality or requiring specialized infrastructure.
Precipitation Methods: Polymer-based precipitation (polyethylene glycol) offers faster processing than ultracentrifugation but introduces distinct challenges. These methods produce broader size distributions indicating contamination with non-exosomal particles. Residual polymer can interfere with downstream applications and introduce cytotoxicity concerns for therapeutic development. Precipitation methods lack ExoDisc's cargo loading capabilities, requiring separate loading protocols if therapeutic applications are intended.
Sonication and Extrusion: These physical disruption methods attempt to permeabilize membranes for cargo loading but cause substantial collateral damage. Research demonstrates that sonication reduces surface marker expression and causes 34.9% particle loss through membrane disruption. Extrusion through narrow-pore membranes creates even more severe damage, with 57% particle loss and compromised tetraspanin marker expression. ExoDisc's gentle tonicity control achieves superior loading efficiency (4.3× and 7.2× improvements respectively) while completely preserving structural and functional integrity.
Therapeutic Applications and Clinical Validation
The published research provides compelling evidence of ExoDisc's utility for therapeutic development through multiple validation experiments demonstrating both cargo loading versatility and functional efficacy.
Diverse Cargo Loading Capability: ExoDisc successfully loaded various therapeutic molecules including doxorubicin hydrochloride (chemotherapeutic), fluorescently-labeled dextrans (3-40 kDa), single-stranded DNA oligonucleotides (10-50 mer), and miRNA-497 (therapeutic RNA). This versatility enables applications across oncology, genetic medicine, and regenerative therapies. The platform's ability to accommodate molecules of different sizes, charges, and chemical properties demonstrates robust applicability to diverse therapeutic development programs.
Functional Therapeutic Validation: Critically, ExoDisc-loaded therapeutics demonstrated superior biological activity compared to conventional loading methods. Doxorubicin-loaded exosomes produced via TC method exhibited 52.1% cytotoxicity against A549 lung cancer cells—substantially higher than sonication (31.1%) or extrusion (25.7%) despite starting from identical sample volumes. This performance gap directly reflects ExoDisc's superior cargo loading efficiency and preservation of exosome functionality.
Three-dimensional tumor spheroid studies validated clinical relevance by demonstrating that ExoDisc-prepared doxorubicin-loaded exosomes achieved 18.1% spheroid size reduction compared to 9.9% with unencapsulated drug—despite equivalent drug concentrations. Fluorescence imaging confirmed 2.2-fold higher drug retention within spheroids when delivered via ExoDisc-prepared exosomes, indicating enhanced cellular uptake and retention. These results suggest potential for improved therapeutic indices in clinical applications where drug delivery efficiency and tumor penetration are critical.
miRNA Therapeutic Development: The platform's utility for nucleic acid therapeutics was validated through miRNA-497 loading experiments. This tumor suppressor miRNA, known to inhibit lung cancer proliferation, was successfully loaded into exosomes at quantities substantially exceeding control methods. Treatment of A549 cells with miRNA-497-loaded exosomes produced significant proliferation inhibition, with ExoDisc-prepared formulations demonstrating superior efficacy compared to control loading methods. This capability supports the growing field of RNA-based therapeutics where exosomes serve as natural delivery vehicles avoiding immunogenicity concerns associated with synthetic nanocarriers.
Quality Control and Reproducibility: Meeting Clinical Standards
Regulatory compliance and quality assurance represent non-negotiable requirements for clinical applications. ExoDisc's design incorporates features supporting rigorous quality control and validation protocols.
Comprehensive Characterization Compatibility: The platform's preserved exosome integrity enables accurate characterization using standard analytical methods. Nanoparticle tracking analysis (NTA) confirms particle concentrations and size distributions. Dynamic light scattering validates size homogeneity. Transmission electron microscopy verifies morphological integrity. Western blotting, ELISA, and flow cytometry characterize surface markers. qRT-PCR validates RNA content. This multi-modal characterization capability ensures comprehensive quality assessment supporting regulatory documentation requirements.
Batch-to-Batch Consistency: Standardized protocols and closed-system processing minimize operator-dependent variables affecting reproducibility. The published research demonstrates consistent performance across multiple independent experiments with low standard deviations, indicating robust reproducibility essential for clinical validation studies and quality control testing.
Contamination Control: The closed-disc design minimizes external contamination risks compared to open-vessel protocols involving multiple transfer steps. This contamination control becomes particularly critical for clinical applications where sample integrity and sterility requirements are stringent. The platform's washing capability effectively removes unencapsulated materials and contaminants, producing clean exosome preparations suitable for therapeutic development.
Cost-Effectiveness and Resource Optimization
Healthcare economics increasingly drive technology adoption decisions. ExoDisc delivers compelling cost-effectiveness through multiple mechanisms that reduce operational expenses while improving outcomes.
Capital Equipment Savings: Eliminating ultracentrifuge requirements removes $30,000-$100,000+ capital equipment barriers, democratizing exosome research access for smaller laboratories and clinical facilities. The platform operates on existing benchtop centrifuges already present in most laboratories, leveraging current infrastructure investments.
Labor Efficiency: Fifteen-minute processing times versus 6-24 hour ultracentrifugation protocols translate to dramatic labor cost reductions. Technicians can process samples within a single work session rather than requiring overnight runs or multi-day protocols. This efficiency enables higher sample throughput with existing staffing, effectively multiplying laboratory capacity without proportional cost increases.
Sample Utilization Optimization: Superior particle recovery (>95% versus 43-80% for other methods) means laboratories extract maximum value from precious clinical samples. When working with limited patient materials or expensive cell cultures, minimizing waste directly impacts project economics and research feasibility. The ability to achieve therapeutic loading goals with less starting material reduces upstream production costs for therapeutic development programs.
Reduced Consumable Costs: The integrated disc platform consolidates multiple process steps that would otherwise require separate vessels, filters, and consumables. This consolidation reduces per-sample consumable expenses while simplifying inventory management and waste disposal requirements.
Future Directions and Expanding Applications
ExoDisc's versatility positions the platform for expanding applications as exosome science advances into new clinical territories.
Personalized Medicine: The platform's rapid processing and preservation of biological function support personalized therapeutic development using patient-derived exosomes as autologous delivery vehicles. This approach bypasses immunogenicity concerns while leveraging exosomes' natural targeting capabilities.
Diagnostic Biomarker Development: The ability to rapidly isolate intact exosomes with preserved molecular cargo supports biomarker discovery and validation studies. Exosomal RNA, protein, and lipid signatures increasingly show promise for early disease detection and treatment monitoring across oncology, neurology, and cardiovascular medicine.
Vaccine Development: Exosomes present attractive vaccine delivery platforms due to inherent immunogenicity and ability to present antigens in physiologically relevant contexts. ExoDisc's cargo loading capabilities support vaccine formulation development with precise antigen loading and quality control.
Regenerative Medicine: Mesenchymal stem cell-derived exosomes demonstrate therapeutic potential in tissue regeneration and immune modulation. ExoDisc enables consistent production and characterization of these therapeutic exosomes at scales supporting clinical translation.
Implementation Considerations for Clinical Laboratories
Laboratories considering ExoDisc adoption should evaluate several practical factors ensuring successful implementation and optimal utilization.
Training and Validation: While ExoDisc operation requires minimal specialized training, laboratories should allocate resources for initial protocol optimization, method validation, and standard operating procedure development. The manufacturer provides comprehensive training support facilitating smooth implementation.
Sample Type Optimization: Different biological matrices (plasma, serum, cell culture supernatants, urine, cerebrospinal fluid) may require protocol modifications for optimal performance. Laboratories should conduct preliminary experiments characterizing performance with their specific sample types, documenting yields, purity metrics, and reproducibility.
Downstream Application Alignment: Selection of cargo loading conditions, washing stringency, and characterization methods should align with intended downstream applications. Therapeutic development programs require more stringent quality control than exploratory research applications. Defining quality specifications upfront guides protocol optimization.
Regulatory Pathway Planning: For laboratories pursuing clinical diagnostic or therapeutic applications, early engagement with regulatory strategy ensures ExoDisc implementation supports compliance requirements. The FDA Class I classification provides a foundation for regulatory documentation, but specific applications may require additional validation studies and quality system documentation.
Clinical and Research Impact: Real-World Outcomes
This research validation of ExoDisc demonstrates tangible improvements in experimental outcomes that translate to accelerated research progress and enhanced clinical potential.
Enhanced Therapeutic Potency: The demonstrated 4.3-7.2-fold improvements in cargo loading efficiency directly translate to more potent therapeutic formulations. This enhanced potency enables lower dosing requirements, potentially improving safety profiles while maintaining efficacy—a critical consideration for therapeutic development programs.
Preserved Biological Function: Complete preservation of surface markers, membrane integrity, and internal components ensures that ExoDisc-isolated exosomes retain native biological activity. This preservation contrasts with physical disruption methods that compromise functionality, yielding vesicles with reduced cellular uptake and biological efficacy. For therapeutic applications where mechanism of action depends on intact exosome function, this preservation represents a fundamental quality advantage.
Reproducible Formulations: The consistency achieved through ExoDisc's standardized processing supports reproducible therapeutic formulation—essential for clinical translation where batch-to-batch consistency determines regulatory approvability. Variable formulations create substantial regulatory barriers; ExoDisc's reproducibility removes this obstacle.
Accelerated Translation: The combined advantages of speed, efficiency, and quality enable accelerated progression from discovery research through preclinical validation to clinical translation. Laboratories can rapidly iterate through formulation optimization cycles, test multiple therapeutic candidates, and generate robust preclinical data supporting clinical applications—all within compressed timelines that improve competitive positioning and accelerate patient access to novel therapies.
Conclusion: A New Standard for Clinical Exosome Technology
ExoDisc represents a transformative advancement in exosome isolation and cargo loading technology, addressing fundamental limitations that have constrained clinical translation of exosome-based applications. The platform delivers unprecedented combination of speed, efficiency, quality, and ease of use—attributes that individually would be valuable but together create a compelling value proposition for clinical laboratories.
The published research validation demonstrates not theoretical potential but actual performance achieving 4.3-7.2-fold improvements in cargo loading while completely preserving exosome integrity. These aren't marginal gains—they represent the performance threshold that makes clinical applications practical rather than aspirational. The 15-minute processing time, benchtop operation, and standard centrifuge compatibility remove infrastructure barriers that have relegated exosome research to specialized centers. ExoDisc democratizes access while simultaneously raising performance standards.
For clinical laboratories evaluating exosome isolation technologies, ExoDisc offers a future-ready platform supporting current research needs while positioning for emerging clinical applications in diagnostics, therapeutics, and personalized medicine. The platform's versatility accommodates diverse sample types, cargo molecules, and applications—providing long-term value as exosome science continues advancing into new clinical territories.
As healthcare increasingly embraces precision medicine and advanced therapeutics, technologies enabling practical implementation of sophisticated approaches become essential infrastructure. ExoDisc delivers this enabling technology, transforming exosome isolation from rate-limiting bottleneck to accelerating capability. Clinical laboratories adopting ExoDisc position themselves at the forefront of exosome science, equipped with tools that turn theoretical potential into clinical reality.
ExoDisc Regulatory and Legal Information
ExoDisc is an FDA Class I in-lab clinical tool designed for the isolation of extracellular vesicles (EVs) from biological samples for use by qualified laboratory professionals in research and clinical laboratory settings.
Regulatory Status: ExoDisc has not been cleared or approved by the FDA for any specific diagnostic, prognostic, or therapeutic application.
Intended Use: ExoDisc is intended solely for research use and professional use in clinical laboratories. It is not intended as the sole determinant for patient diagnosis, treatment, or disease management.
Professional Judgment: Clinical laboratories are responsible for determining the appropriate application of ExoDisc within their validated workflows. All results generated using ExoDisc-isolated extracellular vesicles must be interpreted by qualified healthcare professionals and considered in conjunction with other relevant clinical and laboratory information.
Off-Label and Institutional Use: Use of ExoDisc for applications not specifically cleared or approved by regulators (off-label use), including research protocols, should be conducted in compliance with all applicable institutional review board (IRB) protocols and regulatory requirements.
Performance Variability: Performance characteristics of ExoDisc may vary based on sample type, processing conditions, and the downstream analytical methods employed. It is the responsibility of each user to establish and validate quality control measures, performance characteristics, and workflow suitability for their specific application.
Compliance: Users are responsible for ensuring that the manner in which they use ExoDisc complies with all applicable institutional, local, state, and federal regulations for their intended use and jurisdiction.
Limitation of Liability: The information provided regarding ExoDisc is for informational purposes only and does not constitute medical advice, diagnosis, or treatment recommendations. The manufacturer and distributor expressly disclaim liability for any clinical decisions, patient outcomes, or legal/regulatory consequences arising from the use or misuse of ExoDisc.
Validation Requirement: Individual laboratories remain solely responsible for establishing, validating, and maintaining effective quality control and standard operating procedures when implementing ExoDisc in laboratory or research workflows.
Consult with regulatory affairs specialists and legal counsel regarding compliance for your specific jurisdiction and intended use.