ExoDisc represents a paradigm shift in exosome isolation for clinical settings. Based on the ExoPRISM (Exosome Precipitation by Ionic Strength Modulation) methodology, this platform utilizes electrolyte solutions to fractionally precipitate proteins and extracellular vesicles from biological samples including plasma, serum, urine, and cell culture media. Unlike ultracentrifugation which requires 8-30 hours and specialized equipment, or polymer precipitation methods requiring overnight incubation, ExoDisc delivers complete isolation in under 60 minutes using standard benchtop centrifugation.
The technology achieved over 90% recovery rates across diverse sample types while maintaining exosome functionality, as demonstrated through wound healing assays and downstream molecular analyses. This exceptional performance stems from the method's ability to exploit specific ion effects, causing sequential precipitation that separates proteins from vesicles based on their solubility profiles. The coprecipitated electrolytes are easily washed away, yielding clean exosome preparations suitable for immediate downstream applications.
For clinical laboratories processing patient samples, ExoDisc offers unprecedented scalability. The platform handles volumes ranging from 1 mL to 125 mL with consistent recovery rates, demonstrating linear relationships between starting volumes and exosome yields. This versatility enables laboratories to adapt protocols based on sample availability without compromising quality, a critical advantage when working with precious clinical specimens.
Understanding the Technology: How ExoDisc Works
ExoDisc technology capitalizes on the Hofmeister series and specific ion effects, where ions uniquely influence solution properties including protein stability and solubility. The systematic evaluation of 14 different electrolytes identified ammonium sulfate as optimal for exosome precipitation due to its polyvalent anions and monovalent cations, which effectively modulate ionic strength to induce vesicle aggregation.
The isolation workflow follows an elegant simplicity that belies its sophisticated biochemical foundation:
1. Fractional Precipitation: Adding electrolyte solution to biological samples in controlled increments generates sequential precipitation. Fibrinogen, the least soluble protein, precipitates first at lower concentrations, while albumin remains in solution until higher concentrations are reached. Exosomes precipitate at intermediate concentrations, enabling their separation from contaminating proteins.
2. Low-Speed Centrifugation: Unlike ultracentrifugation at 120,000g for 2-4 hours, ExoDisc utilizes accessible centrifugation speeds of 3,000-10,000g for 10-30 minutes depending on sample volume. This approach eliminates the need for costly ultracentrifuges and reduces mechanical stress on vesicles.
3. Efficient Washing: Coprecipitated electrolytes and residual proteins are removed through ultrafiltration using standard centrifugal filters, with electrochemical impedance spectroscopy confirming negligible salt residue (below 1 mM) in final preparations.
4. Quality Assurance: Cell viability assays demonstrated that trace ammonium sulfate levels maintain over 95% cell viability even at concentrations 10-fold higher than detection limits, confirming biocompatibility for downstream functional studies.
Clinical Workflow Integration: Designed for Real-World Applications
Modern clinical laboratories demand solutions that integrate seamlessly with existing infrastructure while delivering reliable, reproducible results. ExoDisc excels across these critical parameters:
Benchtop Operation Without Specialized Equipment
Traditional ultracentrifugation requires expensive ultracentrifuges, extensive operator training, and processes only 6 samples simultaneously over 8-30 hours. PubMed Central ExoDisc eliminates these constraints by utilizing standard laboratory centrifuges already present in most clinical settings. This accessibility democratizes exosome research and clinical testing, enabling smaller laboratories and point-of-care facilities to perform sophisticated vesicle isolation without capital equipment investments exceeding $100,000.
Reproducibility and Standardization
In repeatability studies involving 24 independent trials, ExoDisc demonstrated coefficient of variation below 3% for exosome recovery, significantly outperforming method-to-method variations reported for ultracentrifugation and commercial kits. This consistency is critical for clinical laboratories requiring validated, reproducible protocols that meet regulatory standards.
Sample Flexibility
ExoDisc successfully isolates exosomes from plasma, serum, urine, and cell culture media across volumes spanning three orders of magnitude (1 mL to 125 mL). This versatility accommodates the diverse sample types encountered in clinical practice, from limited pediatric blood draws to large-volume therapeutic production campaigns.
Preservation of Bioactivity
Functional validation through wound healing assays demonstrated that ExoDisc-isolated exosomes significantly enhanced scratch-wound closure in a dose-dependent manner, outperforming ultracentrifugation-isolated vesicles which showed no significant effect compared to media controls. This preservation of biological function ensures that isolated vesicles retain their native properties for both diagnostic biomarker analysis and potential therapeutic applications.
Ultra-High Yield: Maximizing Diagnostic Research Sensitivity
Exosome yield directly impacts diagnostic research sensitivity—more vesicles enable detection of rare biomarkers critical for early disease diagnosis and monitoring treatment response. ExoDisc delivers industry-leading recovery rates:
Comparative Performance Across Sample Types
For cell culture media, ExoDisc achieved 93% recovery compared to 85% for ultrafiltration, 68% for ExoQuick, and only 48% for ultracentrifugation. RT-PCR analysis revealed ExoDisc-isolated vesicles contained 24-fold more GAPDH mRNA, 26-fold more CD9, 29-fold more CD63, 13-fold more Alix, 8-fold more PSA, 22-fold more PSMA, 15-fold more EpCAM, and 12-fold more EGFR compared to ultracentrifugation.
For clinical plasma samples, these advantages translate to enhanced biomarker detection capability. In plasma spiked with H1975 cell-derived exosomes, ExoDisc enabled detection of EGFR mutations (L858R and T790M) by ddPCR at concentrations as low as 10^7 EVs/mL, while other methods failed to detect mutations at this concentration.
Scalability for Therapeutic Research and QC Applications
Linear relationships between starting volumes and recovered exosome quantities, validated through both sandwich ELISA and RT-PCR across 1-125 mL volumes, confirm ExoDisc's suitability for scaling from diagnostic to therapeutic production. This scalability addresses a critical gap—while tangential flow filtration combined with density gradient ultracentrifugation has been used for therapeutic exosome isolation in clinical trials, these methods require expensive equipment and complex protocols unsuitable for most laboratories.
Purity and Downstream Application Compatibility
Contaminating proteins interfere with mass spectrometry, inflate protein quantification, and confound biomarker analyses. ExoDisc addresses these concerns through optimized precipitation conditions:
Single-step ExoDisc depletes 78% of total plasma proteins while recovering over 90% of exosomes. For applications requiring higher purity, ExoDisc+ combines ionic precipitation with density gradient ultracentrifugation, achieving 97.9% fibrinogen depletion, 91.3% IgG removal, and 99.94% albumin elimination while maintaining 66-71% exosome recovery.
Proteomic analysis identified 530 proteins across ExoDisc+, size-exclusion chromatography, and ultracentrifugation methods, with 190 proteins common to all three, demonstrating comparable protein composition. ExoDisc+ preparations contained 36 of the top 100 Vesiclepedia proteins and 30 of the top 100 Exocarta proteins, confirming enrichment of bona fide exosomal markers.
Clinical Validation: From Bench to Bedside
Clinical validation studies comparing plasma from healthy donors (n=10) versus lung cancer patients (n=10) demonstrated that ExoDisc-isolated exosomes from cancer patients exhibited significantly elevated levels of CD63, EGFR, and Caveolin-1 compared to healthy controls. This differential expression pattern highlights ExoDisc's utility for clinical biomarker discovery and diagnostic development.
The platform's performance characteristics align with key requirements for clinical-scale exosome applications identified in recent comprehensive reviews: rapid processing, ease of use, high yield, high purity, characterization capability, safety, and cost-effectiveness. PubMed
Competitive Landscape: ExoDisc vs. Traditional Methods
The exosome isolation market has evolved significantly, with ultracentrifugation, polymer precipitation (ExoQ****, Total Exosome Isolation Reagent’ Anonymized), size-exclusion chromatography, immunoaffinity capture, and ultrafiltration representing the primary established approaches. Exosome Isolation by Ultracentrifugation and Precipitation: A Comparison of Techniques for Downstream Analyses - PMC +2 Each method presents distinct tradeoffs between yield, purity, processing time, cost, and equipment requirements.
Comparative Performance Table
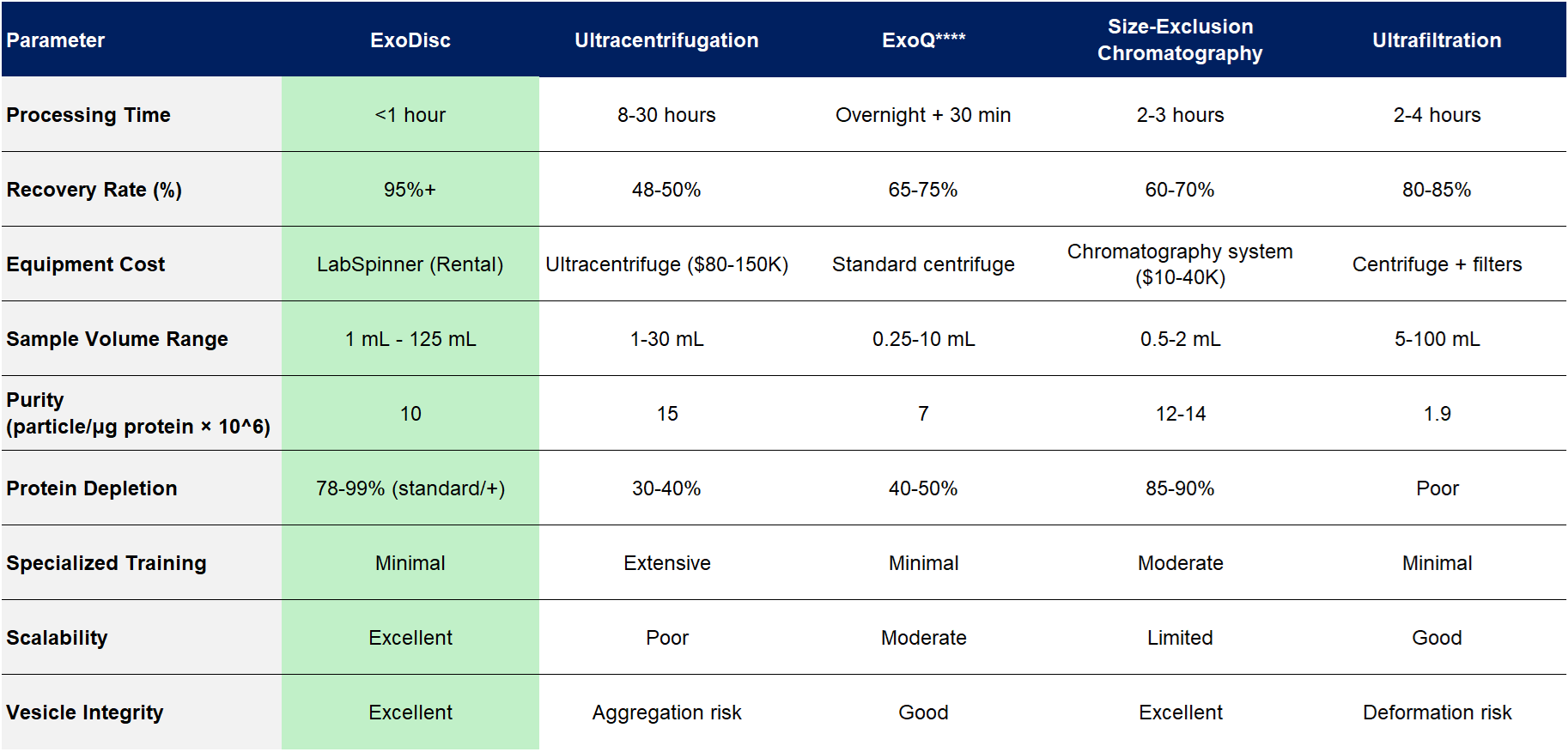
Key Differentiators
Speed Without Sacrifice: ExoDisc's sub-1-hour processing time represents a 10-30-fold reduction compared to ultracentrifugation, enabling same-day results for clinical decision-making. Traditional ultracentrifugation protocols exceeding 10 hours have been identified as a primary barrier to clinical translation and point-of-care testing applications. NCBI
Superior Recovery: ExoDisc's 93% recovery rate nearly doubles ultracentrifugation's 48% yield, providing enhanced sensitivity for rare biomarker detection critical in early cancer diagnosis and treatment monitoring. Comparative studies have demonstrated that precipitation methods can deliver up to 130-fold higher exosome recovery than differential centrifugation when processing clinical volumes. PubMed Central
Cost-Effectiveness: By eliminating the need for ultracentrifuges and reducing processing time, ExoDisc significantly reduces the cost per sample. Commercial precipitation kits like ExoQuick ULTRA emphasize cost savings compared to ultracentrifugation, but require overnight incubation and deliver lower yields than ExoDisc. System Biosciences The rapid turnaround enables higher laboratory throughput, further improving economic efficiency.
Benchtop Accessibility: ExoDisc operates with standard laboratory centrifuges at 3,000-10,000g, compared to ultracentrifugation's requirement for specialized equipment operating at 120,000g. This accessibility is particularly valuable for smaller laboratories, emerging markets, and point-of-care settings where capital equipment costs present barriers to adoption.
Applications in Precision Medicine
The unique performance profile of ExoDisc enables multiple clinical applications:
Liquid Biopsy and Early Diagnosis: Successful detection of EGFR mutations in plasma-derived exosomes at clinically relevant concentrations demonstrates ExoDisc's utility for non-invasive cancer genotyping and treatment selection. The platform's high yield enhances sensitivity for detecting rare circulating tumor-derived exosomes in early-stage disease.
Treatment Monitoring: Differential expression of cancer-associated markers (EGFR, Caveolin-1, CD63) between patient and control exosomes validates ExoDisc for longitudinal monitoring of treatment response and disease progression. Rapid processing enables actionable results within clinical decision-making timeframes.
Biomarker Discovery: Comprehensive proteomic analysis identified over 500 proteins in ExoDisc preparations, with enrichment of exosomal markers and effective depletion of major plasma proteins. This balance between yield and purity creates an optimal platform for discovering novel disease-associated biomarkers.
Research Applications: Beyond diagnostics, ExoDisc supports basic research into exosome biology, drug delivery, and intercellular communication. Preserved biological function demonstrated through wound healing assays confirms that isolated vesicles retain native properties critical for mechanistic studies.
Quality Control and Validation
Robust quality control is essential for clinical implementation. ExoDisc-isolated vesicles have been extensively characterized:
• Size Distribution: Nanoparticle tracking analysis confirms particles in the 40-150 nm size range characteristic of exosomes, with no significant size differences compared to original samples.
• Exosomal Markers: Flow cytometry, Western blotting, and ELISA demonstrate expression of canonical exosomal proteins (CD9, CD63, CD81, TSG101, Alix) and absence of cellular contaminants (calnexin).
• Morphology: Transmission electron microscopy reveals characteristic cup-shaped morphology with intact lipid bilayers, and immunogold labeling confirms surface expression of exosomal markers.
• Functional Validation: Multiple functional assays including wound healing, cell proliferation, and nucleic acid transfer confirm biological activity of isolated vesicles.
________________________________________
Summary: Transforming Clinical Exosome Isolation
ExoDisc represents a significant advancement in clinical exosome isolation technology, delivering the unique combination of rapid processing (<1 hour), ultra-high yields (>95% recovery), benchtop accessibility, and preserved biological function. By leveraging ionic strength modulation, the platform overcomes traditional tradeoffs between speed, yield, and purity while integrating seamlessly into existing clinical workflows.
For laboratories seeking to implement exosome-based diagnostics or expand research capabilities, ExoDisc offers a compelling value proposition: superior performance without specialized equipment, cost-effective operation enabling high-throughput processing, and validated reproducibility meeting regulatory standards for clinical applications.
As precision medicine continues to evolve, exosomes are positioned to play increasingly central roles in non-invasive diagnostics, treatment monitoring, and therapeutic development. ExoDisc provides the foundational technology to translate this promise into clinical reality—delivering the quantity, quality, and speed required for routine clinical implementation.



