Study Overview: ExoDisc Performance in Intraocular EV Therapeutics Development
A landmark comparative study published in Investigative Ophthalmology & Visual Science evaluated ExoDisc against differential ultracentrifugation (dUC) for isolating small extracellular vesicles from human embryonic stem cell-derived retinal pigment epithelium. The research employed orthogonal characterization approaches using multiple devices (NanoSight, ZetaView, and Flow Nanoanalyzer) to rigorously assess yield, purity, scalability, and molecular integrity. Results overwhelmingly favored ExoDisc's tangential flow filtration technology, demonstrating superior performance across all critical parameters essential for clinical translation.
Ultra-High Yield: Maximizing Therapeutic Potential
Clinical exosome therapy demands optimal particle recovery to meet therapeutic dosing requirements. ExoDisc delivered particle yields ranging from 3.13 × 10¹¹ particles/mL across measurement platforms, representing a 3-8 fold increase compared to ultracentrifugation from identical 5 mL input volumes. This dramatic yield enhancement translates directly to:
• Reduced sample input requirements – Achieve target concentrations with significantly less starting material
• Cost efficiency – Maximize exosome recovery from expensive cell culture media
• Therapeutic viability – Meet concentration demands for intraocular injections with limited volume constraints
The platform demonstrated strong correlation (r = 0.98, P = 0.016) between input volume and particle concentration up to 10 mL, with ExoDisc recovering approximately five times more sEVs per milliliter of input compared to conventional methods. For clinical laboratories processing precious biological samples, this efficiency represents a paradigm shift in resource utilization.
Exceptional Purity: Meeting Regulatory Standards
Purity remains paramount for clinical-grade exosome preparations to minimize immunogenic contaminants and ensure therapeutic safety. ExoDisc-isolated exosomes exhibited a particle-to-protein ratio of 3.98 × 10¹⁰ particles/mg protein—5.4-fold higher than ultracentrifugation-derived preparations (7.43 × 10⁹ particles/mg protein, P < 0.0001). This superior purity profile offers:
• Reduced protein contamination – Lower risk of adverse immune reactions
• Consistent batch quality – Enhanced reproducibility for preclinical and clinical studies
• Regulatory compliance readiness – Alignment with cGMP manufacturing requirements
• Preserved microRNA content – Comparable total miRNA concentration while achieving higher purity
Studies consistently show tangential flow filtration surpasses ultracentrifugation in isolating higher yields with better reproducibility, reduced contamination, and preserved EV integrity—critical factors for therapeutic applications PubMed CentralWiley.
Rapid Benchtop Operation: Transforming Laboratory Workflow
Time efficiency directly impacts laboratory throughput and operational costs. ExoDisc completes the entire isolation process in approximately 30 minutes using a compact benchtop centrifuge, compared to 450 minutes required for differential ultracentrifugation. This 15-fold time reduction enables:
• Same-day processing – From sample collection to characterization within hours
• Increased laboratory capacity – Process multiple samples per day
• Reduced labor costs – Minimized hands-on time and operator intervention
• Point-of-care potential – Feasibility for decentralized clinical settings
The automated lab-on-a-disc format eliminates complex multi-step protocols, reducing technical variability and operator-dependent errors that compromise batch consistency.
Seamless Clinical Workflow Integration
ExoDisc's design philosophy prioritizes clinical practicality:
Single-Use Disposable Cartridges
Sterile, pre-packaged ExoDisc cartridges with 20 nm nanoporous membranes eliminate cross-contamination risks and reduce validation burden between runs PubMed Central. Each disposable unit ensures batch-to-batch consistency critical for GMP environments.
Minimal Equipment Footprint
The benchtop centrifuge operates at low g-force values (<500g) compared to ultracentrifugation (>150,000g), eliminating the need for dedicated ultracentrifuge facilities and specialized maintenance PubMed Central.
Scalability for Clinical Manufacturing
ExoDisc demonstrated reliable scalability with input volumes from 1 mL to 10 mL while maintaining high purity, positioning it as a bridge technology from research-scale to clinical-scale production. Tangential flow filtration systems are recognized as suitable for scale-up with GMP compatibility Cytiva.
Quality Control Compatibility
Comprehensive characterization using multiple orthogonal platforms (NanoSight, ZetaView, Flow Nanoanalyzer, TEM, SP-IRIS, and MACSPlex) confirmed ExoDisc-isolated exosomes maintain morphological integrity and surface marker profiles consistent with high-quality EVs.
Competitive Technology Comparison
The table below compares ExoDisc against established exosome isolation methodologies based on published literature:
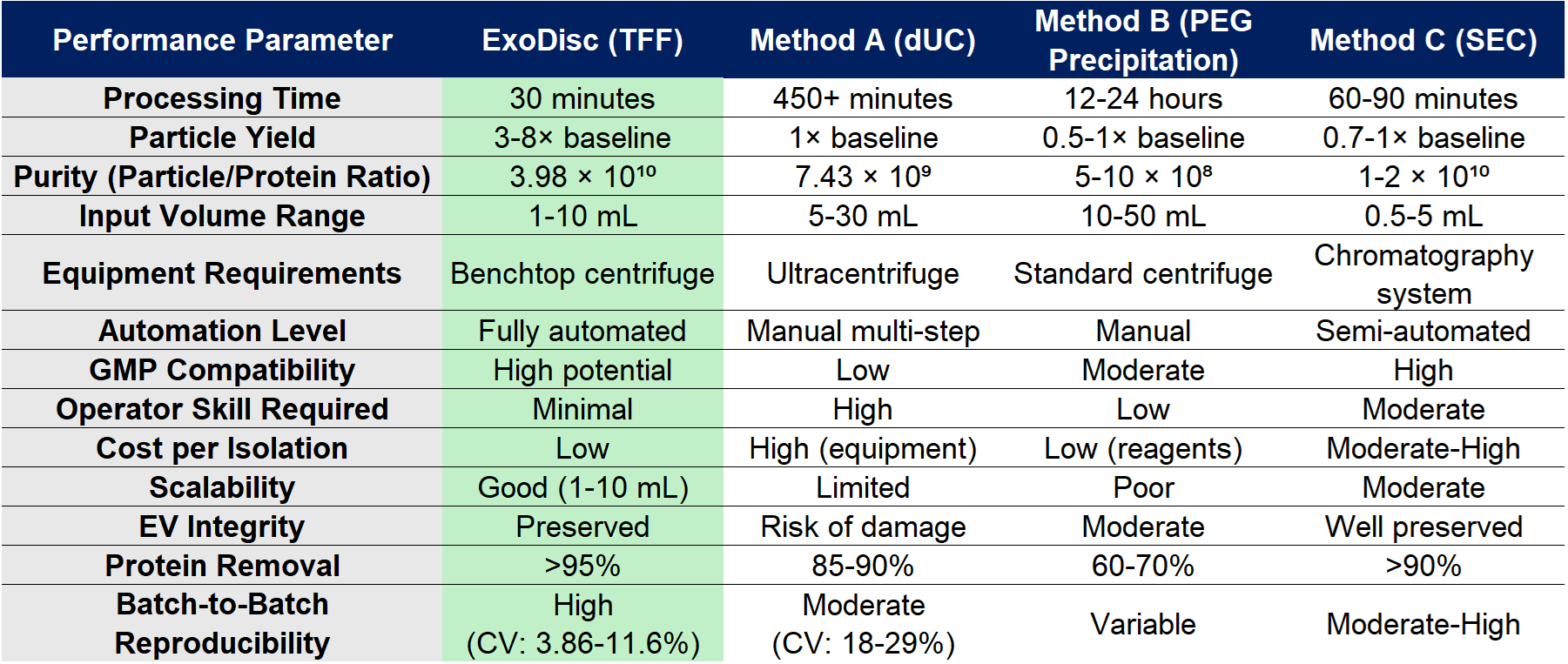
Scientific Validation: Orthogonal Characterization Confirms Quality
The study employed rigorous orthogonal approaches to validate ExoDisc performance, utilizing multiple analytical platforms to establish comprehensive characterization profiles:
Biophysical Characterization
• Transmission electron microscopy confirmed cup-shaped morphology with 30-120 nm size range
• Three independent particle analyzers demonstrated consistent size distributions
• Lower coefficient of variation (3.86-11.6%) compared to ultracentrifugation (18-29%)
Molecular Profiling
Surface marker analysis using SP-IRIS and MACSPlex assays revealed preserved tetraspanin expression patterns (CD9, CD63, CD81) and stem cell-specific markers (SSEA-4, CD133/1, CD29) comparable between ExoDisc and ultracentrifugation methods. This molecular consistency confirms ExoDisc preserves the native biological characteristics essential for therapeutic applications.
Clinical Translation Research Pathway
The development of stem cell-derived intraocular EV therapeutics demands reproducible frameworks for isolation and characterization, particularly given the limited injection volumes and high concentration requirements for intraocular delivery. ExoDisc addresses these challenges through:
Preclinical Development Support
• Consistent batch production for dose-escalation studies
• Reproducible characterization enabling mechanistic investigations
• Sufficient yield for animal efficacy models
Clinical Manufacturing Readiness
The platform's sterile single-use cartridges, automated operation, and GMP-compatible design position ExoDisc as a viable option for Phase I/II clinical trial material production
Regulatory Documentation
Comprehensive analytical validation using multiple orthogonal methods provides robust data packages for regulatory submissions (IND applications)
Clinical Laboratory Advantages: Practical Implementation
Reduced Infrastructure Burden
Eliminate costly ultracentrifuge installations, dedicated clean rooms, and specialized training programs. ExoDisc operates on standard benchtop equipment already present in most clinical laboratories.
Enhanced Laboratory Safety
Low g-force operation (< 500g) reduces equipment hazards and mechanical stress on samples compared to ultracentrifugation protocols requiring >150,000g PubMed Central.
Improved Sample Preservation
Tangential flow filtration's gentle separation principle minimizes shear stress, preserving exosome structural and functional integrity—critical for therapeutic applications PubMed Central.
Quality Assurance Benefits
Single-use disposables eliminate cleaning validation requirements, reduce contamination risks, and simplify batch release testing protocols.
Clinical Research Applications: Expanding Therapeutic Research Horizons
ExoDisc's performance characteristics make it particularly suitable for:
Regenerative Medicine Research
Stem cell-derived exosome therapeutics for retinal diseases, where intraocular injectionvolumes are limited (50-100 μL) requiring ultra-high concentrations
Diagnostic Development Reserach
Liquid biopsy reserach requiring rapid, consistent exosome isolation from clinicalsamples for biomarker discovery and validation
Immunotherapy Enhancement Research
MSC-derived exosomes for immune modulation in autoimmune conditions where batch consistency and purity directly impact therapeutic efficacy study
Personalized Medicine
Point-of-care exosome isolation for patient-specific therapeutic development and monitoring research
Investment in Excellence: Cost-Effectiveness Analysis
Direct Cost Savings
• 15× faster processing reduces labor costs
• Higher yield minimizes expensive cell culture media requirements
• Single-use cartridges eliminate cleaning/validation expenses
• Reduced equipment maintenance compared to ultracentrifuges
Indirect Value Generation
• Increased laboratory throughput enables more clinical samples
• Faster turnaround times accelerate research timelines
• Better reproducibility reduces failed batches and wasted resources
• Enhanced data quality strengthens regulatory submissions
Future-Proofing Your Laboratory
The field of EV therapeutics is rapidly evolving, with growing recognition that diverse isolation technologies are needed to optimize EV preparation for specific applications. ExoDisc positions laboratories at the forefront of this revolution by:
• Enabling participation in cutting-edge clinical trials
• Supporting diverse research applications from diagnostics to therapeutics
• Providing regulatory-ready manufacturing capabilities
• Offering scalability for commercial translation
Conclusion: The ExoDisc Advantage
Published evidence demonstrates tangential flow filtration as a reliable and robust sEV isolation approach that surpasses ultracentrifugation in yield, reproducibility, time, costs, and scalability Wiley. ExoDisc translates these scientific advantages into practical clinical laboratory benefits:
✓ 3-8× higher exosome yield maximizes precious sample utilization
✓ 5.4× superior purity meets stringent therapeutic standards
✓ 15× faster processing transforms laboratory throughput
✓ Benchtop operation eliminates infrastructure barriers
✓ Single-use cartridges ensure batch consistency and safety
✓ GMP-compatible design accelerates clinical translation
✓ Proven performance validated through peer-reviewed research
For clinics developing next-generation exosome therapeutics or diagnostic platforms, ExoDisc represents a strategic R&D investment in quality, efficiency, and clinical readiness. The platform's demonstrated performance in isolating stem cell-derived exosomes for demanding applications like intraocular therapy research underscores its versatility across diverse clinical scenarios.
ExoDisc: Where scientific excellence meets clinical practicality.
________________________________________
This blog post is based on peer-reviewed research published in: Leung J, Pollalis D, Nair GKG, et al. Isolation and characterization of extracellular vesicles through orthogonal approaches for the development of intraocular EV therapy. Invest Ophthalmol Vis Sci. 2024;65(3):6. Additional comparative data compiled from published literature on exosome isolation methodologies.
ExoDisc Regulatory and Legal Information
ExoDisc is an FDA Class I in-lab clinical tool designed for the isolation of extracellular vesicles (EVs) from biological samples for use by qualified laboratory professionals in research and clinical laboratory settings.
Regulatory Status: ExoDisc has not been cleared or approved by the FDA for any specific diagnostic, prognostic, or therapeutic application.
Intended Use: ExoDisc is intended solely for research use and professional use in clinical laboratories. It is not intended as the sole determinant for patient diagnosis, treatment, or disease management.
Professional Judgment: Clinical laboratories are responsible for determining the appropriate application of ExoDisc within their validated workflows. All results generated using ExoDisc-isolated extracellular vesicles must be interpreted by qualified healthcare professionals and considered in conjunction with other relevant clinical and laboratory information.
Off-Label and Institutional Use: Use of ExoDisc for applications not specifically cleared or approved by regulators (off-label use), including research protocols, should be conducted in compliance with all applicable institutional review board (IRB) protocols and regulatory requirements.
Performance Variability: Performance characteristics of ExoDisc may vary based on sample type, processing conditions, and the downstream analytical methods employed. It is the responsibility of each user to establish and validate quality control measures, performance characteristics, and workflow suitability for their specific application.
Compliance: Users are responsible for ensuring that the manner in which they use ExoDisc complies with all applicable institutional, local, state, and federal regulations for their intended use and jurisdiction.
Limitation of Liability: The information provided regarding ExoDisc is for informational purposes only and does not constitute medical advice, diagnosis, or treatment recommendations. The manufacturer and distributor expressly disclaim liability for any clinical decisions, patient outcomes, or legal/regulatory consequences arising from the use or misuse of ExoDisc.
Validation Requirement: Individual laboratories remain solely responsible for establishing, validating, and maintaining effective quality control and standard operating procedures when implementing ExoDisc in laboratory or research workflows.
Consult with regulatory affairs specialists and legal counsel regarding compliance for your specific jurisdiction and intended use.



