Automated Excellence in Exosome Isolation
Published in Theranostics (2019), this groundbreaking study validates ExoDisc-B as a fully integrated centrifugal microfluidic device capable of automatic, rapid, and efficient isolation of extracellular vesicles directly from whole blood. The research demonstrates that ExoDisc isolates at least 10-fold more exosomes than ultracentrifugation with approximately 30-fold higher mRNA content, all within 40 minutes. With capture efficiency exceeding 75% from blood volumes as low as 30 µL, the platform successfully differentiated 43 prostate cancer patients from 30 healthy donors through ELISA analysis of cancer-related proteins. The study encompassed comprehensive performance validation including nanoparticle tracking analysis, protein quantification, RNA content assessment, and longitudinal monitoring of tumor progression in mouse xenograft models—confirming ExoDisc's capacity to provide intact, high-quality exosomes suitable for downstream clinical analysis.
The Clinical Challenge: Why Traditional Exosome Isolation Falls Short
Extracellular vesicles hold immense promise as biomarkers for cancer diagnosis and monitoring, yet their clinical adoption has been severely constrained by the absence of simple, robust isolation methods. Current ultracentrifugation protocols demand multiple time-consuming, labor-intensive high-speed centrifugation steps that not only consume 6+ hours but may also compromise exosome integrity through high centrifugal forces.
Blood plasma's complexity—containing cells, lipoprotein particles, proteins, and high viscosity—creates significant technical hurdles. During ultracentrifugation, protein aggregates co-precipitate with exosomes and lipoprotein particles of similar size and density, resulting in poor quantitative and qualitative performance during downstream analysis.
For clinical laboratories seeking to integrate liquid biopsy workflows, these limitations translate to:
• Extended turnaround times incompatible with clinical decision-making timelines
• Substantial sample volume requirements that limit serial monitoring capabilities
• Inconsistent results that compromise diagnostic confidence
• Specialized equipment and technical expertise requirements
• Limited scalability for routine clinical testing
ExoDisc Platform Technology: Engineering Excellence for Clinical Workflows
ExoDisc employs an innovative dual-filtration architecture operating through sequential, tangential flow-filtration principles. The platform comprises a built-in chamber facilitating plasma separation and two nanoporous filters—one (600 nm) removing larger particles and another (100 nm AAO membrane) enriching exosomes.
The tangential flow-filtration mechanism represents a critical design innovation. By positioning membrane filters such that flow direction remains perpendicular to centrifugal pumping force, ExoDisc operates at relatively low g-force values (less than 500 g) while achieving sequential filtration. This approach minimizes membrane fouling—a persistent challenge in conventional filtration methods—while preserving exosome structural integrity.
The entire process integrates five individually addressable diaphragm valves enabling automatic control of sequential sample transfer. This automation encompasses plasma separation, pre-filtration, exosome enrichment, washing, and elution—eliminating manual intervention points where contamination or user error might compromise results.
Unmatched Performance Metrics: The Numbers That Matter
The ExoDisc platform delivers quantifiable performance advantages verified through rigorous comparative analysis against ultracentrifugation:
Yield Superiority: ExoDisc isolates at least an order of magnitude higher numbers of exosomes than ultracentrifugation. CD9-CD81 sandwich ELISA confirmed capture efficiencies of approximately 76% for ExoDisc with 100nm AAO membrane (D100) compared to just 15% for ultracentrifugation.
Molecular Content Preservation: ExoDisc-isolated exosomes contained approximately 30-fold higher mRNA concentration than ultracentrifugation-isolated samples, as confirmed by RT-PCR analysis of GAPDH, CD9, CD63, and CD81 mRNA. This substantial difference in recovered nucleic acid content directly impacts downstream molecular profiling applications.
Purity Excellence: ExoDisc-prepared exosomes demonstrated significantly lower albumin contamination (0.61 ± 0.07 µg/mL) compared to ultracentrifugation (9.03 ± 0.78 µg/mL), as measured by both ELISA and Western blot. This approximately 15-fold reduction in protein contamination enhances the accuracy of subsequent proteomic and functional analyses.
Minimal Volume Capability: ExoDisc successfully isolates exosomes from whole blood volumes as low as 30 µL—a game-changing capability for serial monitoring, pediatric applications, and research involving small animal models where blood volume conservation is critical.
Speed Advantage: The complete exosome enrichment process requires 10-40 minutes depending on sample volume (30-600 µL), compared to 6+ hours for ultracentrifugation protocols. This time efficiency enables same-day turnaround compatible with clinical workflows.
Clinical Validation: From Bench to Diagnostic Insights
The platform's clinical utility was rigorously validated through two distinct study paradigms:
Longitudinal Monitoring in Xenograft Models: ExoDisc enabled continuous real-time monitoring of tumor progression in mouse xenograft models over 13 weeks using minimal plasma volumes (less than 30 µL per week). ELISA measurements of five protein markers (CD9, CD81, PSMA, EGFR, and HSP90) demonstrated significant increases in cancer-associated markers as early as two weeks post-inoculation. This proof-of-concept directly demonstrates ExoDisc's value for therapy monitoring and disease progression tracking.
Human Clinical Sample Differentiation: Plasma-derived exosomes from 43 prostate cancer patients and 30 healthy donors were isolated using ExoDisc and analyzed for multiple protein markers via ELISA. All tested markers—including EV-specific proteins (CD9, CD63, CD81) and cancer-associated proteins (PSA, PSMA, EpCAM, EGFR1, HSP90)—successfully differentiated cancer patients from healthy donors with statistical significance.
Among tested markers, HSP90 demonstrated the highest diagnostic performance with 86% sensitivity, 90% specificity, 88% accuracy, and area under ROC curve of 0.92. While this finding requires validation in larger patient cohorts, it illustrates ExoDisc's capacity to support biomarker discovery and validation workflows.
Seamless Clinical Workflow Integration
ExoDisc addresses practical clinical laboratory needs through several design considerations:
Benchtop Operation: The compact spinning system requires minimal laboratory footprint, operating as a point-of-care device without specialized infrastructure requirements.
Minimal Training Requirements: Single manual sample injection initiates the fully automated process, enabling operation by any personnel without special training.
Immediate Downstream Compatibility: Isolated exosomes can be directly used for conventional analyses including nanoparticle tracking analysis, ELISA, PCR, Western blot, and mass spectrometry without additional purification steps.
Sample Flexibility: The platform accommodates both fresh whole blood (ExoDisc-B) and pre-stored plasma samples (ExoDisc-P), providing operational flexibility for diverse laboratory workflows.
Reproducibility: Testing across multiple devices demonstrated coefficient of variation below 7%, ensuring consistent performance critical for clinical diagnostics.
Competitive Landscape: How ExoDisc Outperforms Alternative Technologies
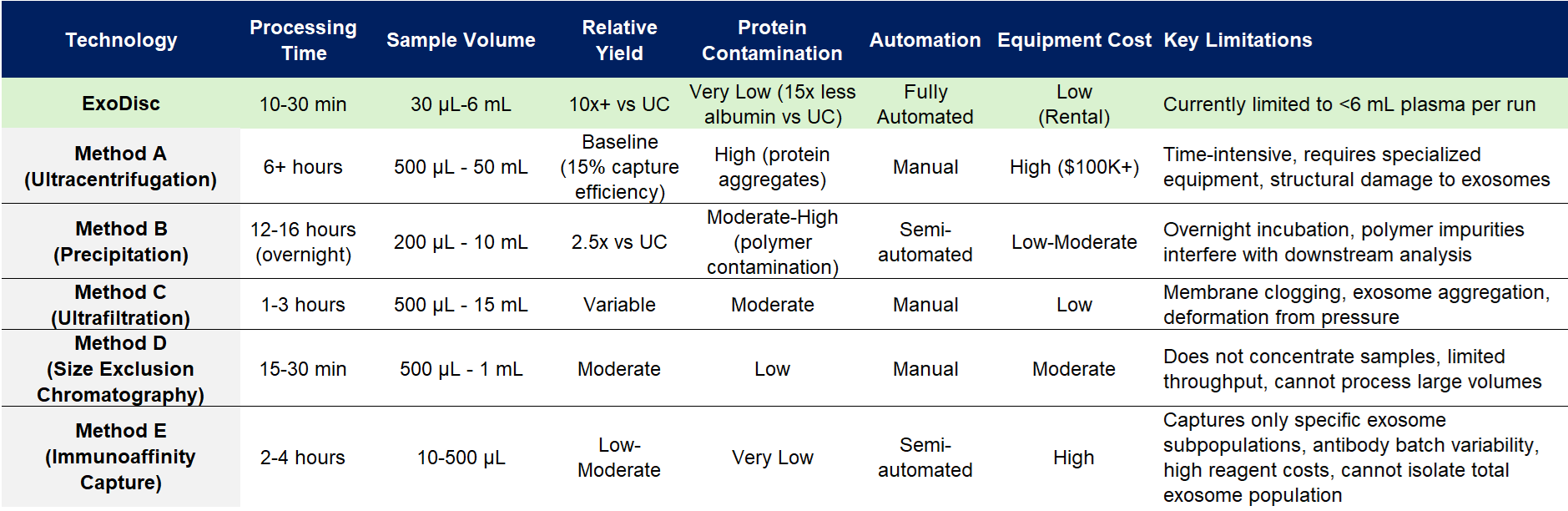
The ExoDisc Difference: Why Clinical Laboratories Choose This Platform
1. Time Is Clinical Value: Where precipitation methods require 6x less time than ultracentrifugation PubMed Central, ExoDisc completes the entire process in 10-40 minutes—enabling same-day diagnostic workflows previously impossible with conventional methods.
2. Quality Without Compromise: The 30-fold increase in recovered mRNA content directly translates to enhanced sensitivity for molecular diagnostics and biomarker validation studies.
3. Minimal Volume, Maximum Information: The 30 µL minimum blood requirement enables applications previously constrained by sample availability—pediatric diagnostics, serial monitoring protocols, and small animal research.
4. Reproducible Results Build Clinical Confidence: Low inter-device variability (CV <7%) ensures consistent performance across multiple laboratory sites—essential for multi-center clinical trials and standardized diagnostic protocols.
5. Workflow Simplicity Accelerates Adoption: Single-step manual injection followed by complete automation removes technical barriers to implementation, accelerating laboratory onboarding and reducing operator-dependent variability.
Application Horizons: Expanding Clinical Possibilities
Cancer Diagnosis and Monitoring: The validated differentiation of cancer patients from healthy donors demonstrates ExoDisc's potential for non-invasive cancer screening and disease surveillance.
Therapy Response Monitoring: The demonstrated capability for longitudinal monitoring with minimal blood volumes positions ExoDisc as an ideal platform for tracking treatment response and detecting minimal residual disease.
Biomarker Discovery: High-purity, high-yield exosome preparations enhance statistical power in biomarker validation studies, potentially accelerating identification of clinically actionable targets.
Precision Medicine: Rapid turnaround times enable real-time molecular profiling to guide therapeutic decision-making—a cornerstone of precision oncology approaches.
Future Perspectives: The Evolution of Liquid Biopsy
While ExoDisc provides exosomes in substantially higher yield and purity compared to ultracentrifugation, isolated preparations still contain mixtures of exosomes with different sizes and subsets as well as some non-exosome contaminants such as low-density lipoprotein. Ongoing platform refinements continue addressing these technical challenges to further enhance preparation purity.
Although the current design limits processing of plasma samples larger than 1 mL per disc, the platform's modular architecture enables parallel processing for larger sample volumes when required. For clinical laboratories, this capacity aligns well with typical diagnostic sample volumes while maintaining rapid turnaround.
The proof-of-concept demonstrations with prostate cancer provide a foundation for expansion into other cancer types and disease states. As the platform undergoes continued clinical validation across diverse patient populations and cancer types, its utility for therapy monitoring and drug-resistance prediction will be more fully established.
Conclusion: Enabling the Liquid Biopsy Revolution
ExoDisc represents more than incremental improvement in exosome isolation methodology—it constitutes a paradigm shift enabling practical clinical implementation of liquid biopsy approaches. By delivering automated, rapid, high-yield exosome enrichment from minimal blood volumes with benchtop convenience, the platform removes critical barriers that have constrained clinical translation of exosome-based technologies.
For clinical laboratories, ExoDisc offers a compelling value proposition: transform 6-hour manual protocols into 30-minute automated workflows while simultaneously improving yield by 10-fold and mRNA recovery by 30-fold. This combination of speed, quality, and operational simplicity positions ExoDisc as an enabling technology for next-generation precision technology.
ExoDisc Regulatory and Legal Information
ExoDisc is an FDA Class I in-lab clinical tool designed for the isolation of extracellular vesicles (EVs) from biological samples for use by qualified laboratory professionals in research and clinical laboratory settings.
Regulatory Status: ExoDisc has not been cleared or approved by the FDA for any specific diagnostic, prognostic, or therapeutic application.
Intended Use: ExoDisc is intended solely for research use and professional use in clinical laboratories. It is not intended as the sole determinant for patient diagnosis, treatment, or disease management.
Professional Judgment: Clinical laboratories are responsible for determining the appropriate application of ExoDisc within their validated workflows. All results generated using ExoDisc-isolated extracellular vesicles must be interpreted by qualified healthcare professionals and considered in conjunction with other relevant clinical and laboratory information.
Off-Label and Institutional Use: Use of ExoDisc for applications not specifically cleared or approved by regulators (off-label use), including research protocols, should be conducted in compliance with all applicable institutional review board (IRB) protocols and regulatory requirements.
Performance Variability: Performance characteristics of ExoDisc may vary based on sample type, processing conditions, and the downstream analytical methods employed. It is the responsibility of each user to establish and validate quality control measures, performance characteristics, and workflow suitability for their specific application.
Compliance: Users are responsible for ensuring that the manner in which they use ExoDisc complies with all applicable institutional, local, state, and federal regulations for their intended use and jurisdiction.
Limitation of Liability: The information provided regarding ExoDisc is for informational purposes only and does not constitute medical advice, diagnosis, or treatment recommendations. The manufacturer and distributor expressly disclaim liability for any clinical decisions, patient outcomes, or legal/regulatory consequences arising from the use or misuse of ExoDisc.
Validation Requirement: Individual laboratories remain solely responsible for establishing, validating, and maintaining effective quality control and standard operating procedures when implementing ExoDisc in laboratory or research workflows.
Consult with regulatory affairs specialists and legal counsel regarding compliance for your specific jurisdiction and intended use.